Press releases
| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
| Su | Mo | Tu | We | Th | Fr | Sa |
|---|---|---|---|---|---|---|
-
Jul 7, 2021New online portal effectively aggregates, customizes and streamlines purchasing of essential reagents to accelerate bold research and clinical innovation
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced the commercial release of a new bdbiosciences.com web and eCommerce site, an entirely...
-
Jun 30, 2021BD also announced plans to host an Investor Day
BD (Becton, Dickinson and Company) (NYSE:BDX) announced today that it will report its financial results for the third quarter of fiscal year 2021 on Thursday, August 5, 2021. BD will issue a press...
-
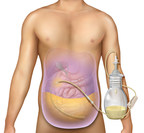
Jun 23, 2021PeritX™ Peritoneal Catheter System is the first and only at-home system indicated for malignant and non-malignant ascites drainage in the United States.
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for...
-
Jun 22, 2021
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today issued its 2020 Sustainability Report, highlighting significant achievements in environmental,...
-
Jun 11, 2021
USA Swimming, the national governing body for the sport of swimming in the United States, and BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, have...
-
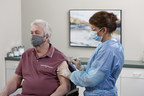
Jun 8, 2021Company Now Supplying More Than 40 Countries for Pandemic Vaccination Campaigns
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced that it has received pandemic orders for needles and syringes totaling 2 billion...
-
Jun 2, 2021Becomes the First Medical Technology Company Authorized as a CVE Numbering Authority
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced that it has become the first medical technology company authorized as a Common...
-
May 26, 2021
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced that it will present at the following upcoming virtual investor conferences during the...
-
May 25, 2021BD Surgiphor™ sterile wound irrigation system delivers simplified sterility and helps hospitals comply with national and international guidelines[1,2]
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, announced today the availability of BD Surgiphor™ Sterile Wound Irrigation System, the first and only...
-
May 18, 2021New plant will produce drug delivery devices, employ 150 people in 2024 and up to 600 when full operations start in 2030
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, will build a €165 million ($200 million USD) high-tech manufacturing facility in the city of...
-
May 13, 2021BD Onclarity™ HPV Assay CE Marked for Self-Collection; Available on BD Viper™ LT and BD COR™ Systems
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced the industry's first self-collection claim for HPV screening has been CE marked to the...
-
May 6, 2021Standalone Diabetes Care business expected to leverage position as global leader in insulin delivery and expand presence in large and high-growth diabetes market
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced its board of directors has unanimously authorized management to proceed with a plan to...
-
May 6, 2021- Second fiscal quarter revenues of $4.9 billion grew 15.4% on a reported basis. On a currency-neutral basis, revenues increased 12.2%.
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today reported quarterly revenues of $4.9 billion for the second fiscal quarter ended March 31, 2021....
-
Apr 29, 2021
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced that it will present at the following upcoming virtual investor healthcare conferences...
-
Apr 29, 2021
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, announced today that enrollment has begun and the first patients have been treated in the post-market...
-
Apr 28, 2021John DeFord to join newly formed external Scientific Advisory Board
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced the appointment of Elizabeth McCombs as executive vice president and chief technology...
-
Apr 27, 2021
The Board of Directors of BD (Becton, Dickinson and Company) (NYSE: BDX) has declared a quarterly dividend of $0.83 per common share, payable on June 30, 2021 to holders of record on June 9, 2021....
-
Apr 27, 2021
CerTest Biotec, along with BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced the VIASURE SARS-CoV-2 (N1 + N2) Real Time PCR Detection Kit...
-
Apr 26, 2021
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced it has submitted a 510(k) premarket notification to the U.S. Food and Drug...
-
Apr 23, 2021
USA Track & Field (USATF) and BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, have partnered to provide rapid COVID-19 testing for USATF athletes at...
-
Apr 22, 2021As part of its 2030+ sustainability goals, BD pledges carbon neutrality by 2040 across direct operations
In recognition of Earth Day, BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, reinforces its commitment to Advancing the world of health™ by affirming...
-
Apr 20, 2021
BD (Becton, Dickinson and Company), a leading global medical technology company, is voluntarily recalling specified lots of the ChloraPrep™ Hi-Lite Orange™ 26 mL Applicator (2% w/w...
-
Apr 20, 2021Company continues its commitment to expanding health care access through its long-standing support of UNICEF USA and their efforts to eliminate Maternal and Neonatal Tetanus
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced a $1 million commitment over five years to UNICEF USA in support of UNICEF's work to...
-
Apr 14, 2021Clearance supported by clinical data from a feasibility study with favorable primary patency rates observed out to 180 days
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced it has received 510(k) clearance from the U.S. Food and Drug Administration (FDA) for...
-
Apr 13, 2021WHO Rapid Communication cites high accuracy for the detection of TB, rifampicin and isoniazid resistance on respiratory samples
BD (Becton, Dickinson and Company) (NYSE: BDX), a leading global medical technology company, today announced that its BD MAX™ Molecular Multi-Drug Resistant Tuberculosis (MDR-TB) Assay was...